News Story
The Internet of Bio-Nano Things

Stock image of a wearable device concept
University of Maryland (UMD) researchers are one step closer to bridging the gap between microelectronics and biological systems, a critical obstacle to transforming today’s smart devices – such as watches or fitness bands – into potentially life-saving technology. Their latest findings were published this week in Nature Nanotechnology.
Once a Jetsons-era pipe dream, smart devices are now part of a wearables market that could grow to nearly $64 billion by 2024, according to a recent GlobalData report. Today, trending wearable devices collect a wealth of data in order to prompt wearers to exercise, snap and send photos, or even improve their posture.
But, what if your fitness monitor or smart watch could detect an active virus in your body – such as COVID-19 – and alert you in real time? Or, what if a diabetes patient could tap similar technology to administer a dose of insulin as soon as their blood glucose level spikes?
"Devices that freely exchange information between the electronic and biological worlds would represent a completely new societal paradigm," said William E. Bentley, UMD Fischell Department of Bioengineering (BIOE) professor, director of UMD’s Robert E. Fischell Institute for Biomedical Devices, and the project’s principal investigator. "It has only been about 60 years since the implantable pacemaker and defibrillator proved what devices could achieve by electronically stimulating ion currents. Imagine what we could do by transferring all the knowledge contained in our molecular space, by tapping into and controlling molecules such as glucose, hormones, DNA, proteins, or polysaccharides in addition to ions."
Despite rapid advances in wearable technology, tech developers face a major roadblock to making such devices a reality.
That’s because today’s microelectronic devices process information using materials such as silicon, gold, or chemicals, and an energy source that provides electrons. But, free electrons do not exist in biological systems. As such, there remains a major gap between microelectronics and the biological world.
Nearly three years ago, Bentley, his Fischell Institute and UMD Institute for Bioscience and Biotechnology Research (IBBR) colleague, Gregory F. Payne, and their respective teams published research on a loophole they discovered.
In biological systems, there already exists a small class of molecules capable of shuttling electrons. These molecules, known as “redox” molecules, can transport electrons to any location. To do this, redox molecules must first undergo a series of chemical reactions – oxidation or reduction reactions – to transport electrons to the intended target.
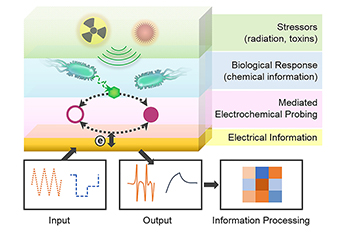
Building on this process, Bentley’s and Payne’s teams joined forces with synthetic biology experts BIOE alumna Jessica Terrell (Ph.D. ‘15) and her colleagues at the U.S. Army Research Lab, and BIOE alumna Tanya Tschirhart (Ph.D. ‘14) and her colleagues at the U.S. Naval Research Lab. Together, they worked to create a microbial network they could "plug into" an external electronic system to interrogate and control biological function in real time. The collaborative team crafted an electronic system and a community of engineered microbial cells to create an electronically controlled biological local area network, dubbed a BioLAN.
In the computer realm, a LAN consists of a collection of devices connected together in a physical location. In a similar way, the team’s BioLAN employs a network of engineered cells to convert information delivered via an electronic input into a biological response. In turn, that biological response conveys information across a microbial network in order to carry out network functions.
One such function is verification that the electronic signal was successfully translated to carry out a biological response. To make this verification possible, the team created a terminal electronic output that confirms that the original “message” was delivered to the appropriate target, similar to how a “read receipt” functions for email.
In addition, the BioLAN carries out a second critical function that involves initiating production and delivery of a therapeutic. The team demonstrated that their BioLAN successfully instructed engineered bacterial cells to create granulocyte macrophage colony stimulating factor (GMCSF), a therapeutic proposed for the treatment of Crohn’s disease and several immunological disorders.
One of the critical components of this system is the “living electrode” the team created by binding specially engineered cells to the surface of a gold electrode. This living electrode allows the team to facilitate the redox reaction needed to translate an electronic signal into information that can be read by a biological system. In this way, the team created a hybrid system with electronically programmed and tracked – yet, biologically executed – functions, a major step in bridging the gap between electronics and biology.
“We are developing an array of tools – including electrogenetics, using electronic signals to actuate gene expression – that will eventually enable seamless information transfer from electronics to biology and back," Bentley said. "Some have referred to such a future as the Internet of Bio-Nano Things. We think that, in addition to making 'smart' devices even smarter, these innovations will bring to life new ways of storing data, making computations, and more. Where human-made systems take advantage of the integration of biological components, there are likely to be opportunities where we use electronics to guide or control biological processes.”
This latter approach could be used to alter the production of foods and medicines, control the delivery of medicine such as from a wearable or ingested microelectronic device, or even control complex microbiological environments such as the microbiome in the gastrointestinal tract.
"If we have the right tools, creative entrepreneurs will be sure to capitalize on the technology," Bentley said.
University of Maryland (UMD) Fischell Department of Bioengineering (BIOE) alumni Jessica L. Terrell (Ph.D. ‘15; U.S. Army Research Laboratory [ARL]) and Tanya Tschirhart (Ph.D. ‘14; U.S. Naval Research Laboratory [NRL]) were first authors of the Nature Nanotechnology paper. In addition to William E. Bentley and Gregory F. Payne, the following also contributed to the paper: Justin P. Jahnke (ARL), BIOE alum Kristina Stephens (Ph.D. ‘20; UMD Robert E. Fischell Institute for Biomedical Devices [Fischell Institute]; UMD Institute for Bioscience and Biotechnology Research [IBBR]), Yi Liu (BIOE; IBBR), Hong Dong (ARL), Margaret M. Hurley (ARL), BIOE alum Maria Pozo (B.S. ‘19), BIOE alum Ryan McKay (Ph.D. ‘18; Fischell Institute; IBBR), Chen Yu Tsao (BIOE, Fischell Institute, IBBR), BIOE alum Hsuan-Chen Wu (Ph.D. ‘12; IBBR), Gary Vora (NRL), and Dimitra N. Stratis-Cullum (ARL). In addition to his BIOE and Fischell Institute appointments, Bentley is affiliated with the UMD Department of Chemical and Biomolecular Engineering and IBBR.
This work received funding support from the U.S. Defense Threat Reduction Agency, the National Science Foundation, the National Institutes of Health, and the Office of Naval Research. This work was also supported by the Office of the Under Secretary of Defense for Research and Engineering through the Applied Research for Advancement of S&T Priorities Program on Synthetic Biology for Military Environments.
Full text of the Nature Nanotechnology paper, titled “Bioelectronic control of a microbial community using surface-assembled electrogenetic cells to route signals,” is available online.
Published March 30, 2021